Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 -Ga
-Ga O
O (001)
(001)Tsai, Y. H.*; Kobata, Masaaki; Fukuda, Tatsuo; Tanida, Hajime; Kobayashi, Toru; Yamashita, Yoshiyuki*
Applied Physics Letters, 124(11), p.112105_1 - 112105_5, 2024/03
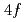 states in the electronic structure of the intermediate-valence superconductor CeIr
states in the electronic structure of the intermediate-valence superconductor CeIr
Fujimori, Shinichi; Kawasaki, Ikuto; Takeda, Yukiharu; Yamagami, Hiroshi; Sasabe, Norimasa*; Sato, Yoshiki*; Shimizu, Yusei*; Nakamura, Ai*; Maruya, A.*; Homma, Yoshiya*; et al.
Electronic Structure (Internet), 5(4), p.045009_1 - 045009_7, 2023/11
 Al
Al ; Impact of the U
; Impact of the U 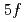 states on the electronic structure of UPd
states on the electronic structure of UPd Al
Al
Fujimori, Shinichi; Takeda, Yukiharu; Yamagami, Hiroshi; Posp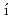
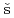 il, J.*; Yamamoto, Etsuji; Haga, Yoshinori
il, J.*; Yamamoto, Etsuji; Haga, Yoshinori
Physical Review B, 105(11), p.115128_1 - 115128_6, 2022/03
Times Cited Count:0 Percentile:0(Materials Science, Multidisciplinary)Baba, Yuji; Shimoyama, Iwao
Photon Factory Activity Report 2016, 2 Pages, 2017/00
In order to elucidate the adsorption states of radioactive Sr-90 in soil, chemical bonding states of non-radioactive strontium adsorbed on layered oxide (mica) have been investigated by X-ray photoelectron spectroscopy (XPS) and X-ray absorption near edge structure (XANES) spectroscopy. Since the number of atoms in radioactive Sr-90 is extremely small, the XPS and XANES were measured under total reflection condition of the incident X-rays. The detection limit in total reflection XPS was about 150 pg/cm , which corresponds to 300 Bq of Sr-90. The Sr 2p
, which corresponds to 300 Bq of Sr-90. The Sr 2p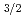 core-level energy in XPS shifted to lower energy with the decrease in the thickness of Sr layer. Also, the Sr 2p
core-level energy in XPS shifted to lower energy with the decrease in the thickness of Sr layer. Also, the Sr 2p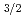
 Sr 4d
Sr 4d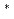 resonance energy in XANES shifts to lower energy with the decrease in the thickness. On the basis of a simple point charge model, it was elucidated that the chemical bond between Sr and mica surface becomes ionic with the decrease in the adsorbed amount of strontium.
resonance energy in XANES shifts to lower energy with the decrease in the thickness. On the basis of a simple point charge model, it was elucidated that the chemical bond between Sr and mica surface becomes ionic with the decrease in the adsorbed amount of strontium.
Okane, Tetsuo; Kobata, Masaaki; Sato, Isamu*; Kobayashi, Keisuke*; Osaka, Masahiko; Yamagami, Hiroshi
Nuclear Engineering and Design, 297, p.251 - 256, 2016/02
Times Cited Count:2 Percentile:19.71(Nuclear Science & Technology)Baba, Yuji; Shimoyama, Iwao; Hirao, Norie; Izumi, Toshinori
e-Journal of Surface Science and Nanotechnology (Internet), 13, p.417 - 421, 2015/09
Times Cited Count:1The interaction between alkali metals and oxides has attracted much attention as heterogeneous catalysis, chemical reaction promoters, and high intensity electron emitter. Also the interaction of cesium and oxides has become important subject, because radioactive cesium trapped in minerals such as clay and soil may cause health risks. In the present study, we analyzed chemical states of ultra-trace amount of cesium on oxide surfaces by total reflection X-ray photoelectron spectroscopy (TR-XPS) exited by synchrotron radiation. For the adsorbed cesium thicker than 0.01 layer, cesium is weakly bound with oxide through Van-der-Waals force. On the other hand, for ultra-thin layer about 0.002 layer, the chemical bond changes to covalent bond. It is suggested that this change in the chemical bonding state is one of the reasons why radioactive cesium is hard to be released from minerals.
Takakuwa, Yuji*; Ogawa, Shuichi*; Ishizuka, Shinji*; Yoshigoe, Akitaka; Teraoka, Yuden
Shokubai, 47(5), p.352 - 357, 2005/08
Oxidation reactions of Ti(0001) surfaces were monitored by real-time photoelectron spectroscopy with high brilliant synchrotron radiation and He-I resonance line. An oxygen adsorption model and ultra-thin oxide layers formation processes were reviewed on the basis of time evolutions of the adsorbed oxygen amount, oxidation states, electronic states and the work function, which were obtained from photoelectron spectra for Ti 2p and O 1s core levels as well as the valence band.
Kameo, Yutaka; Nakashima, Mikio; Hirabayashi, Takakuni*
Journal of Nuclear Science and Technology, 41(9), p.919 - 924, 2004/09
Times Cited Count:12 Percentile:61.53(Nuclear Science & Technology)A new laser decontamination method utilizing a gel made from a sodium silicate solution and an acid was developed for removing radioactive nuclide incorporated into a surface oxide layer on metal waste. Decontamination tests were carried out using both simulated contaminated samples and pipe specimens cut from the primary coolant system of the Japan Power Demonstration Reactor. In the case of surface oxide layer consisting mainly of iron oxide, more than 99% of surface radioactivities were removed after two to three decontamination runs. In order to ascertain the role of laser irradiation on chemical reactions, chemical states of O and Fe in the oxide layer before and after decontamination were analyzed by X-ray photoelectron spectroscopy. It was found that the oxide layer was dissolved into the acid-containing gel, and the reaction was extensively promoted by the laser irradiation.
Sasaki, Masayoshi*; Morimoto, Yasutomi*; Kimura, Hiromi*; Takahashi, Koji; Sakamoto, Keishi; Imai, Tsuyoshi; Okuno, Kenji*
Journal of Nuclear Materials, 329-333(Part1), p.899 - 903, 2004/08
A CVD diamond has been the reference material of a torus widow for a rf heating system in a fusion reactor. Since the window is in the circumstance of tritium, helium and radio activated dust, it is important to elucidate the effect of ions on chemical structure of the diamond, existing states of tritium and tritium inventory. Polycrystalline CVD diamond disks(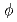 =10.0mm, t=0.21mm) used in this study are the same grade as rf windows. After sputtering the surface with 1.0 keV Ar
=10.0mm, t=0.21mm) used in this study are the same grade as rf windows. After sputtering the surface with 1.0 keV Ar to remove oxygen impurity, the sample was irradiated with deuterium (D
to remove oxygen impurity, the sample was irradiated with deuterium (D
 ) or helium ions (He
) or helium ions (He ) at an angle of 0 degrees to the surface normal. The irradiation energies of deuterium and helium are 0.25 keV D
) at an angle of 0 degrees to the surface normal. The irradiation energies of deuterium and helium are 0.25 keV D and 0.45 keV He
and 0.45 keV He , respectively. The structural change of the irradiated sample was measured by X-ray Photoelectron Spectroscopy (XPS) technique. The C1s peak shift toward lower binding energy side was observed when deuterium ions irradiated. This result indicates that the diamond changes to amorphous carbon due to formation of C-D bond.
, respectively. The structural change of the irradiated sample was measured by X-ray Photoelectron Spectroscopy (XPS) technique. The C1s peak shift toward lower binding energy side was observed when deuterium ions irradiated. This result indicates that the diamond changes to amorphous carbon due to formation of C-D bond.
Takakuwa, Yuji*; Ishizuka, Shinji*; Yoshigoe, Akitaka; Teraoka, Yuden; Mizuno, Yoshiyuki*; Tonda, Hideki*; Homma, Teiichi*
Hyomen Kagaku, 24(8), p.500 - 508, 2003/08
Temperature dependence of the initial oxidation kinetics of Ti(0001) surface was investigated by low energy electron diffraction (LEED) and real-time photoelectron spectroscopy. The LEED observation revealed that the oxide layers grew epitaxially. From the oxygen uptake curve, it was clarified that oxigen diffusion was significantly enhanced by surface temperature and the oxide layers thickness reached to 7 nm at 400 C. From Ti-2p photoelectron spectroscopy, TiO
C. From Ti-2p photoelectron spectroscopy, TiO was predominant at the subsurface while TiO and Ti
was predominant at the subsurface while TiO and Ti O
O components were also observed at the interface.
components were also observed at the interface.
Takakuwa, Yuji*; Ishizuka, Shinji*; Yoshigoe, Akitaka; Teraoka, Yuden; Yamauchi, Yasuhiro*; Mizuno, Yoshiyuki*; Tonda, Hideki*; Homma, Teiichi*
Applied Surface Science, 216(1-4), p.395 - 401, 2003/06
Times Cited Count:19 Percentile:64.2(Chemistry, Physical)Real-time in-situ observation using photoelectron spectroscopy for elementary processes of Ti(0001) oxidation by O molecules has been performed at the surface reaction analysis apparatus installed at the BL23SU in the SPring-8. And the real-time observation has been also performed by RHEED-AES methods at Tohoku University. The partial pressure region of oxygen was from 2x10
molecules has been performed at the surface reaction analysis apparatus installed at the BL23SU in the SPring-8. And the real-time observation has been also performed by RHEED-AES methods at Tohoku University. The partial pressure region of oxygen was from 2x10 Torr to 8x10
Torr to 8x10 Torr. The surface temperature was 473 K and 673 K. The variation from clean Ti surface toward TiO
Torr. The surface temperature was 473 K and 673 K. The variation from clean Ti surface toward TiO was comfirmed by observation of Ti-2p and O-1s photoelectron spectra. Reflected electron intensity and O-KLL Auger electron intensity oscillated in the RHEED-AES measurements. These facts revealed that the surface morphological change of the oxidized Ti(0001) surface was associated not only with a disappearance of the surface metallic layer but also with a change of the oxidation state.
was comfirmed by observation of Ti-2p and O-1s photoelectron spectra. Reflected electron intensity and O-KLL Auger electron intensity oscillated in the RHEED-AES measurements. These facts revealed that the surface morphological change of the oxidized Ti(0001) surface was associated not only with a disappearance of the surface metallic layer but also with a change of the oxidation state.
Takakuwa, Yuji*; Ishizuka, Shinji*; Yoshigoe, Akitaka; Teraoka, Yuden; Mizuno, Yoshiyuki*; Tonda, Hideki*; Homma, Teiichi*
Nuclear Instruments and Methods in Physics Research B, 200, p.376 - 381, 2003/01
Times Cited Count:10 Percentile:56.43(Instruments & Instrumentation)no abstracts in English
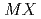 (
(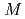 =Pt, Pd;
=Pt, Pd; 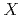 =P, As, Sb)
=P, As, Sb)Iwasaki, T.*; Sekiyama, Akira*; Yamasaki, Atsushi*; Okazaki, Makoto*; Kadono, Koji*; Utsunomiya, Hiroshi*; Imada, Shin*; Saito, Yuji; Muro, Takayuki*; Matsushita, Tomohiro*; et al.
Physical Review B, 65(19), p.195109_1 - 195109_9, 2002/05
Times Cited Count:24 Percentile:71.84(Materials Science, Multidisciplinary)no abstracts in English
 and V
and V O
O after rare-gas sputtering
after rare-gas sputteringSasaki, Teikichi; Baba, Yuji; Yamamoto, Hiroyuki; Sasase, Masato*; *
Surface and Interface Analysis, 20, p.682 - 686, 1993/00
Times Cited Count:4 Percentile:21.43(Chemistry, Physical)no abstracts in English
Baba, Yuji; Yamamoto, Hiroyuki; Sasaki, Teikichi
Nuclear Instruments and Methods in Physics Research B, 66, p.424 - 432, 1992/00
Times Cited Count:7 Percentile:60.99(Instruments & Instrumentation)no abstracts in English
Baba, Yuji; Yamamoto, Hiroyuki; Sasaki, Teikichi
Nuclear Instruments and Methods in Physics Research B, 63, p.391 - 394, 1992/00
Times Cited Count:7 Percentile:60.99(Instruments & Instrumentation)no abstracts in English

;
Journal of Nuclear Materials, 152, p.295 - 300, 1988/00
Times Cited Count:4 Percentile:46.81(Materials Science, Multidisciplinary)no abstracts in English
Baba, Yuji
JAERI 1304, 76 Pages, 1987/02
no abstracts in English
 N
N and SiO
and SiO by energetic hydrogens
by energetic hydrogens;
Journal of Nuclear Materials, 138, p.145 - 148, 1986/00
Times Cited Count:5 Percentile:85.2(Materials Science, Multidisciplinary)no abstracts in English
;
Journal of Nuclear Materials, 138, p.149 - 151, 1986/00
Times Cited Count:1 Percentile:56.32(Materials Science, Multidisciplinary)no abstracts in English